by Jonathan Vaught, Ph.D.
CEO, QiPath
New scientific opportunities, rising complexity, and increasing regulatory demands are making traditional methods of risk management unsustainable for pharmaceutical organizations. Our industry is experiencing:
- An explosion of advanced biologics and other novel modalities.
- New levels of operational complexity brought about by these modalities—personalized modalities, globalized manufacturing, sensitive supply chains, decentralized trials, and rapidly expanding data streams.
- Regulators that expect clearer, more proactive oversight to ensure continuous compliance with strict regulations to ensure patient safety and product efficacy.
Maintaining proper Quality Systems management in this environment requires a framework that can focus on completing complex actions, preventing costly errors, and driving continuous, dynamic process improvement and operational excellence. All in real time.
![]() Traditional risk management was built for a simpler time. Today’s risk methods are unable to adequately govern the dynamic, evolving systems required for innovative therapies being developed. As the gap between static risk methods and evolving manufacturing systems grows, so will delays, surprises, and compliance burdens. Regulators have noticed. They’re expecting clearer, more proactive oversight, not retrospective documentation exercises.
Traditional risk management was built for a simpler time. Today’s risk methods are unable to adequately govern the dynamic, evolving systems required for innovative therapies being developed. As the gap between static risk methods and evolving manufacturing systems grows, so will delays, surprises, and compliance burdens. Regulators have noticed. They’re expecting clearer, more proactive oversight, not retrospective documentation exercises.
Closing that gap requires risk frameworks that are adaptive (updating as science, processes, and partners change), continuous (always current rather than tied to periodic reviews), learnable (improving through human insight and operational data), and process-linked (grounded in how work actually happens, not how it’s documented).
This is the foundation of the Adaptive Risk Intelligence (ARI) model.
What Adaptive Risk Intelligence Is—and Isn’t
ARI doesn’t replace traditional risk management. It modernizes the approach by integrating artificial intelligence with human expertise and operational data, so organizations can prevent failures rather than just documenting them.
Building an effective ARI model requires rethinking three assumptions that most organizations take for granted.
Risk Lives in Processes, Not Documents
Most risk assessments capture vulnerabilities in the form of lists, categories, or narratives—disconnected from the processes that generate them. But risk doesn’t originate in documents. It originates in behaviors, handoffs, constraints, timing, and decision points.
This disconnect becomes increasingly costly as pipelines rely more on sensitive biological materials and complex workflows. Risks described in static documents stop reflecting how work actually unfolds. Lessons learned at one site can’t scale to others. And AI tools can’t learn because risks aren’t mapped to real process behavior.
A process-centric foundation can change this. By linking vulnerabilities to where they actually originate, organizations gain richer insight, clearer accountability, and a structure that enables meaningful AI learning. Without this foundation, even sophisticated AI will generate generic recommendations disconnected from operational reality.
Human-in-the-Loop Means Contribution, Not Just Approval
AI in pharmaceutical risk management remains at the early stage. Most current implementations position humans as gatekeepers who are reviewing and approving AI-generated suggestions before they’re acted upon. That’s not human-in-the-loop. That’s a checkpoint.
In a mature ARI model, people become continuous contributors. Frontline staff, process engineers, and quality professionals feed insight into the system as a natural byproduct of their routine work rather than additional tasks requiring specialized expertise or workflow disruption.
This requires tools designed for minimal friction: capturing insight passively where possible, converting tacit operational knowledge into structured signals, and enabling contribution through small, embedded interactions rather than formal assessment exercises.
Done well, this approach does more than enrich the risk model. It strengthens AI validation by grounding the system in real operations rather than idealized process descriptions. And it supports a fundamental shift from a reactive assessment to predictive understanding of emerging risks.
AI Needs Operational Context to Create Value
Generative AI can summarize documents and propose potential risks. But without operational context, it reflects only general knowledge and not the specific behaviors of your sites, vendors, equipment, or processes.
The real value of ARI emerges when AI is progressively paired with internal operational signals such as QMS events, CTMS activity, MES patterns, batch records, and site performance trends. This integration moves risk intelligence from descriptive (“what the process should do”) to predictive (“what the process is likely to do next”).
Organizations don’t need deep data integration to start. A process-centric structure and systematic human input form the foundation. Operational data can be layered in over time, accelerating learning and amplifying value as the system matures.
From Framework to Outcomes
When these elements come together—process-centric structure, continuous human contribution, and AI informed by operational signals—the results compound.
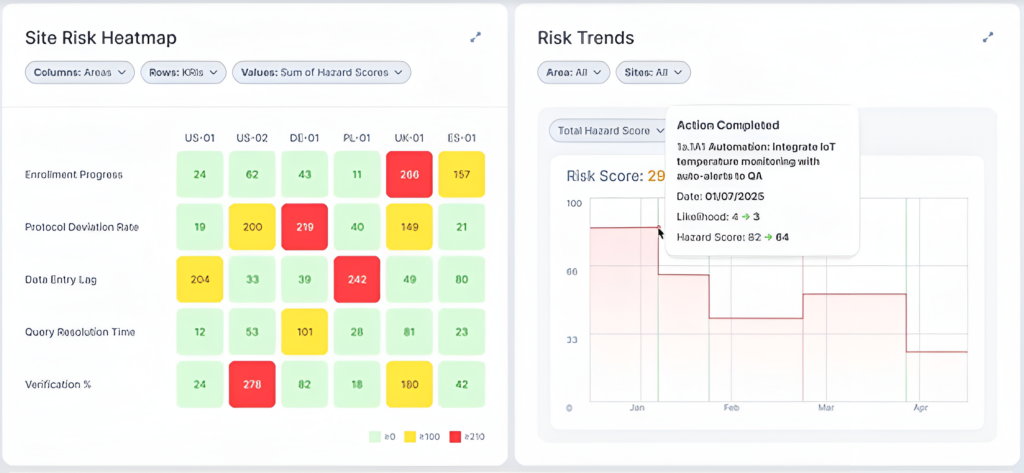
The above dashboard shows how teams can compare risk indicators at a glance (left) while tracking how specific interventions (such as integrating IoT temperature monitoring) reduce hazard scores over time (right). This visibility transforms risk management from periodic review to continuous oversight.
Earlier signal detection means small shifts get flagged before they escalate into batch failures, protocol deviations, or extended timelines. Process-based risk analysis enables mitigation strategies that align with how work actually happens, driving timely execution rather than theoretical controls.
Leaders gain clearer visibility into risk trajectories across programs, sites, partners, and CMOs. This supports better prioritization, smarter resource allocation, and fewer reactive fire drills.
Perhaps most importantly, compliance becomes a natural outcome rather than an administrative burden. Traceability, version control, and defensible logic are built into the structure. Part 11 and GxP expectations are met as an inherent byproduct of a well-governed system instead of being reconstructed after the fact (and in an urgent manner) for auditors.
The organizations that adapt their risk frameworks now won’t just avoid problems. They’ll operate with a level of confidence and control that static methods simply cannot provide.
 About the Author:
About the Author: